Julius Robert von Mayer was largely ignored by other professionals in the area. Not long after Robert Clausius developed his theories about heat and work into a concise version of the Second Law of Thermodynamics concerning the increase of entropy in natural systems.

Pdf The Third Law Of Thermodynamics Or An Absolute Definition For Entropy Part 1 The Origin And Applications In Thermodynamics
Boyles Law states that in order for the temperature and pressure in hell to stay the same the ratio of the mass of souls and volume needs to stay constant.
Who wrote the laws of thermodynamics. 1 an open system allows mass and energy to flow into or out of the system. Julius Robert Von Mayer the First in History to Formulate the First Law of Thermodynamics. By 1860 as formalized in the works of scientists such as Rudolf Clausius and William Thomson what are now known as the first and second laws were established.
This marks the start of thermodynamics as a modern science. Who wrote the law of thermodynamics. Who wrote the first law of thermodynamics.
As expected they prove to be in. One such scientist was Sadi Carnot the father of thermodynamics who in 1824 published Reflections on the Motive Power of Fire a discourse on heat power and engine efficiency. The first law of thermodynamics is the physical law which states that the total energy of a system and its surroundings remain constant.
Heilbronn November 25 1814 - March 20 1878. Who wrote the three laws of thermodynamics. Which best expresses the first law of thermodynamics.
The laws of thermodynamics would proceed as usual. Credit for the law of Conservation of Energy is given to the German scientists Robert Mayer 1814-1878 and Hermann von Helmholtz 1821-1894. Ad ber 7 Millionen englischsprachige Bcher.
As illustrated in Fig. The second law also states that the changes in the entropy in the universe can never be negative. But the idea of energy as a unique quantity conserved during ALL physical interactions wasnt stated until the development of thermodynamics in the mid-1800s.
Ad ber 7 Millionen englischsprachige Bcher. Around 1850 Rudolf Clausius and William Thomson Kelvin stated both the First Law - that total energy is conserved - and the Second Law of Thermodynamics. The Laws of Thermodynamics which science itself recognizes in its explanations of the phenomena in the Universe were written by the Chief Engineer cf.
So was born the First Law of Thermodynamics the principle of conservation of energy. We begin with the first law of thermodynamics applied to an open thermodynamic system. Ralph H Fowler gave the term Zeroth law of thermodynamics in 1935.
Who wrote the first law of thermodynamics. The Second Law was originally formulated in terms of the fact that heat does not spontaneously flow from a. The law is also known as the law of conservation of energy which states energy can transform from one form into another but can neither be created nor destroyed within an isolated systemPerpetual motion machines of the.
The Second Law of Thermodynamics The second law of thermodynamics was first expressed in stated form by the German physicist Rudolf Clausius 1822-1888. However although not all Creation models are in harmony with the scientific evidence one would expect the true Creation model to be in keeping with the evidence. Julius Robert von Mayer was a German physician and physicist and one of the founders of the principles of thermodynamics.
According to the first law of thermodynamics energy cannot be created or destroyed only transformed from one form to another. D ont hold your breath waiting for a perpetual motion machine. The first law of thermodynamics and investing risk.
The Second Law of Thermodynamics states that the state of entropy of the entire universe as an isolated system will always increase over time. 2nd Law of Thermodynamics. One if hell is expanding at a slower rate than the rate t which souls enter hell then the temperature and pressure in hell will increase exponentially until all hell breaks loose.
There are two possible conditions. The short answer. He observed that it is not possible to construct an engine whose sole effect is the extraction of heat from a heat source at a single temperature and the conversion of this heat completely into mechanical work.
In 1935 Fowler was reading the texts of two physicists Meghnad Saha and B N Srivastava and he came up on the text that Every physical quantity must be measurable in numeric terms. Many have claimed over the centuries they can beat the law of the conservation of energy. And we would still view the low entropy direction in time as the past and the high entropy direction as the future.
Physics as a Foreign Language. Does the first law of thermodynamics apply to open systems.
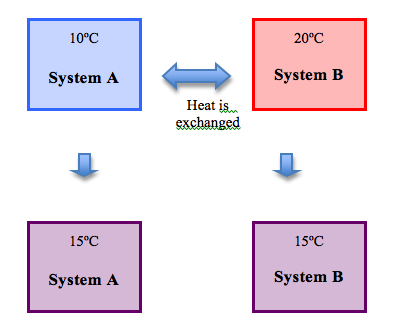
0th Law Of Thermodynamics Chemistry Libretexts

Pdf The Second Law Of Thermodynamics And Entropy
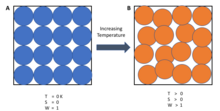
Laws Of Thermodynamics Wikipedia

Pdf Student Understanding Of The First Law Of Thermodynamics Relating Work To The Adiabatic Compression Of An Ideal Gas

First Law Of Thermodynamics Wikipedia
Why Is Zeroth Law Of Thermodynamics Named Zeroth Law Quora

Pdf Historical Observations On Laws Of Thermodynamics

First Law Of Thermodynamics Universe Today
Why The Is Zeroth Law Of Thermodynamics Called Zeroth Law Why Did They Didn T Start The Counting From One Quora

Pdf Conservation Of Energy Readings On The Origins Of The First Law Of Thermodynamics Part Ii

The Fourth Law Of Thermodynamics Steepest Entropy Ascent Philosophical Transactions Of The Royal Society A Mathematical Physical And Engineering Sciences
Why Is Zeroth Law Of Thermodynamics Named Zeroth Law Quora
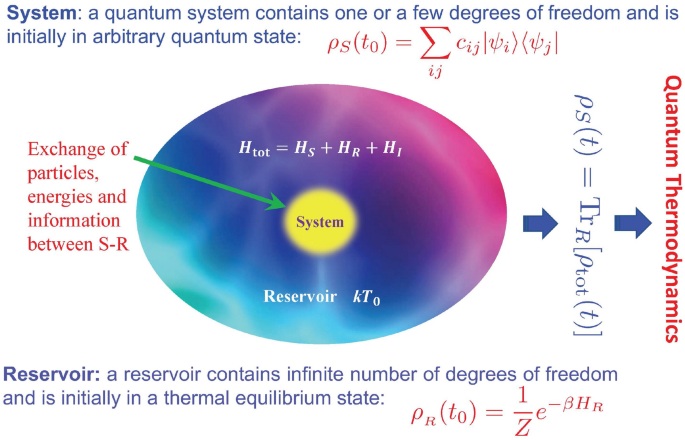
Quantum Thermodynamics Of Single Particle Systems Scientific Reports

The Laws Of Thermodynamics A Very Short Introduction Atkins Peter 9780199572199 Amazon Com Books

The Social Implications Of The Second Law Of Thermodynamics Gene Veith


Post a Comment
Post a Comment